Service hotline
+86 18518316054
 Current location : Home page > Resources > Papers > Monodisperse SnS2 Nanosheets for High-Performance Photocatalytic Hydrogen Generation
Current location : Home page > Resources > Papers > Monodisperse SnS2 Nanosheets for High-Performance Photocatalytic Hydrogen Generation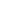
ABSTRACT:
Graphene-like two-dimensional layered materials have attracted quite a lot of interest because of their sizable band gaps and potential applications. In this work, monodisperse tin disulfide (SnS2) nanosheets were successfully prepared by a simple solvothermal procedure in the presence of polyvinylpyrrolidone (PVP). Large PVP molecules absorbing on (001) facets of SnS2 would inhibit crystal growth along [001] orientation and protect the product from agglomeration. The obtained SnS2 nanosheets have diameters of ca. 0.8−1 μm and thicknesses of ca. 22 nm. Different experiment parameters were carried out to investigate the transformation of phase and morphology. The formation mechanism was proposed according to the time-dependent experiments. SnS2 nanosheets exhibit high photocatalytic H2 evolution activity of 1.06 mmol h−1 g−1 under simulated sunlight irradiation, much higher than that of SnS2 with different morphologies and P25-TiO2. Moreover, the as-obtained SnS2nanosheets show excellent photoelectrochemical response performance in visible-light region.
EXPERIMENTAL SECTION
Photocatalytic Activity of SnS2 NSs. Photocatalytic hydrogen evolution from water was carried out in CEL-SPH2Nphotocatalytic activity evaluation system (Beijing Au-light, China). The reactor used in the photocatalytic process was a cylindrical quartz vessel with height of 10 cm and diameter of 7 cm. The area of the light irradiation is approximately 38.5 cm2 . The system was well sealed with Dow Corning high vacuum grease. A 300W Xe lamp (CEL-HXF 300, Beijing Au-light, China, I = 20 A) was employed as simulated sunlight source. In a typical photocatalytic experiment, 20 mg of catalyst powder was dispersed into 100 mL 10 vol % of methanol solution. 0.1 M Na2S and 0.1 M Na2SO3 were added as sacrificial agents. Before catalytic reaction, the whole system was vacuumized using a mechanical pump. Typically, 0.4 mL of gas was extracted each hour and analyzed using online gas chromatograph (GC7890 II, TECHCOMP, China, N2 carrier). The amount of hydrogen production was calculated according to the fitted standard curve. The used catalyst was recycled by centrifuging the suspensions after catalytic reaction, and washed with deionized water for several times. The obtained catalyst was then dried in air at 60 °C before the next cycle of reaction.
The photoelectrochemical response of the samples at 0.8 V versus SCE electrode under 300 W Xe lamp illumination (λ > 420 nm). (B) The electrochemical impedance spectra. Z′ and Z″ represent the real and imaginary parts of the impedance, while the solid lines were fitted by ZSimpWin software by the equivalent circuits. (a) SnS2 NSs; (b) the sample obtained without PVP added; and (c) the sample prepared using H2O as solvent with 0.5 g PVP.
CONCLUSIONS
The large-size SnS2 NSs with diameters of 0.8−1 μm and thicknesses of ca. 22 nm were successfully synthesized via a one-step solvothermal method. Experiments of different reaction parameters reveal that orthorhombic SnS microflowers were first formed in the initial step. With the increase of reaction time, SnS would be transferred to be hexagonal SnS2. As a steric stabilizer or capping agent, PVP molecules would preferentially absorb on the (001) facet of SnS2 nuclei, inhibitcrystal growth along [001] orientation, and protect the product from aggregation. The as-prepared SnS2 NSs exhibit good dispersity over a long time standing. The formation mechanism was proposed to undergo a nucleation−dissolution−recrystallization−growth procedure. SnS2 NSs demonstrate excellent photocatalytic water splitting performance. The average H2production rate was detected to be 1.06 mmol h−1 g−1 , which is much higher than that of SnS2 of different morphologies and commercial P25-TiO2. Furthermore, the visible-light response ability is revealed to be outstanding compared with irregular SnS2 nanoplates, indicating the potential application of SnS2NSs in photodetector and photovoltaic fields. This approach might provide a valuable guidance to the family of MX2 materials.